Decoding Hydrogen Spillover: Unraveling the Dual-Site Relay-Guided C-C Coupling Mechanism in CO2 Electroreduction
Tanghong Zheng1, Mengmeng Xu1, Haoyu Zhang1, Xinran Qiu1, Youyong Li2, Qingfeng Xu1(徐庆锋)*, Jianmei Lu1(路建美)*
1College of Chemistry, Chemical Engineering and Materials Science, Collaborative Innovation Center of Suzhou Nano Science and Technology, Soochow University, Suzhou, Jiangsu 215123, China
2Institute of Functional Nano & Soft Materials (FUNSOM), Jiangsu Key Laboratory for Carbon-Based Functional Materials & Devices, Soochow University, Suzhou, Jiangsu 215123, China
Inorg. Chem.2025, 64, 19026–19035
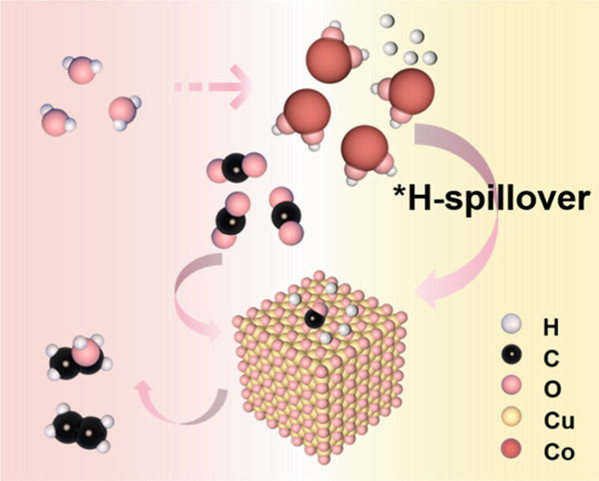
Abstract: The electrochemical reduction of CO2 into valuable C2+ products presents a sustainable and efficient strategy for the utilization of CO2 and long-term renewable energy storage. Yet, enhancing the efficiency of the electrocatalytic CO2 reduction reaction (eCO2RR) in aqueous systems remains challenging due to the difficulty in activating both CO2 and H2O molecules. In this study, we focus on water activation generating reactive hydrogen species (*H) to boost C2+ product selectivity. Toward this target, we develop a series of bimetallic Cu-based catalysts with modulated Co loadings to form dual active sites. The optimized 0.24% Co–Cu2O catalyst demonstrated exceptional performance, achieving a faradaic efficiency (FE) of 76.1% for C2+ products at a high-current density of 400 mA cm–2. Remarkably, it maintained stable FEC2+ values of >70% for over 24 h under continuous operation, indicating robust operational stability. Experimental studies and DFT calculations revealed that Co sites promote water activation to generate *H. The generated *H subsequently spills over to Cu sites, modulating the *H coverage on Cu surfaces, which enhances the conversion of *CO to *CHO intermediates. Subsequently, the asymmetric coupling between *CHO and *CO at the Cu sites generates C2+ products. This site-segregation strategy optimizes the selectivity and FE for the CO2-to-C2+ conversion.
Article information: //doi.org/10.1021/acs.inorgchem.5c03152