Bench-Stable Carbamoylzinc Pivalates for Modular Access to Amides, Ureas, and Thiocarbamates
Donghao Luo, Liangjie Ruan, Jiayi Xue, Zicong Jiang, Changrui Nie, Ting Zeng, Laiman Luo, Jie Li(李杰)*
State Key Laboratory of Bioinspired Interfacial Materials Science, MOE Key Laboratory of Geriatric Diseases and Immunology, Key Laboratory of Organic Synthesis of Jiangsu Province, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, China
J. Am. Chem. Soc.2025, 147, 32238–32248
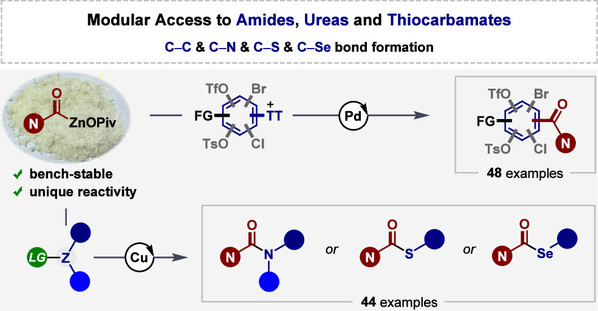
Abstract: Amide units and relative carbonyl families such as ureas and their derivatives are central important backbones of numerous compounds with activities of relevance to biology or medicinal chemistry. Driven by their prevalence, a general technology that enables sustainable amide-unit synthesis should afford new opportunities for chemical innovation. Generally, stoichiometric quantities of activating reagents, (tri)phosgene and its derivatives, or CO are commonly used in the literature to construct such scaffolds, which represent the drawbacks of these approaches. As such, we herein report a new series of salt-stabilized carbamoylzinc pivalates prepared from the corresponding formamides through C–H metalation in the presence of TMPLi (TMP = 2,2,6,6-tetramethylpiperidyl) and Zn(OPiv)2. These carbamoylzinc pivalates are obtained as powders with high air and moisture stability under ambient conditions. They also show excellent reactivity in palladium-catalyzed C–S-selective Negishi carbamoylation and copper-catalyzed electrophilic amination, thiolation, and selenolation, thus unlocking a new platform to access functionalized amides, unsymmetric ureas, thiocarbamates, and selenocarbamates through C–H zincation and C–C or C–heteroatom coupling reactions in a sustainable manner. Finally, late-stage modification of drug-like molecules and direct synthesis of drugs showcase the potential of this protocol in medicinal chemistry.
Article information: //doi.org/10.1021/jacs.5c12107