Electrochemical Synthesis of Hydrogen Peroxide Enabled by Tri-Coordinated Cobalt Sites in Silicate-1 Zeolite
Guanghua Yu1, Yingying Jin1, Ye Zhou1, Jing Xia3(夏静)*, Xiangmin Meng3, Guoliang Dai4(戴国梁)*, Jiong Wang1,2(王炯)*
1Innovation Center for Chemical Science, College of Chemistry, Chemical Engineering and Materials Science, Soochow University,Suzhou 215006, China
2Jiangsu Key Laboratory of Advanced Negative Carbon Technologies, Soochow University, Suzhou 215123, China
3Key Laboratory of Photochemical Conversion and Optoelectronic Materials, Technical Institute of Physics and Chemistry Chinese Academy of Sciences, Beijing 100190, China
4School of Chemistry and Life Sciences, Suzhou University of Science and Technology, Suzhou 215009, China
Angew. Chem. Int. Ed. 2025, 64, e202506390
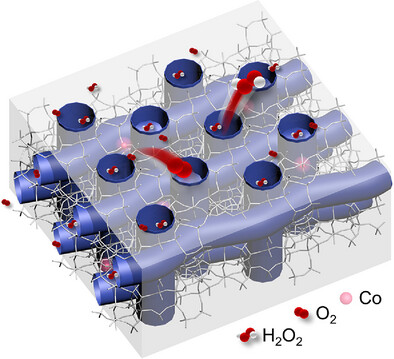
Abstract:Electrochemical synthesis of hydrogen peroxide (H2O2) via metal-catalyzed two-electron oxygen reduction reaction (2e− ORR) emerges as a sustainable alternative to the traditional anthraquinone method. However, metal catalysts are commonly supported by graphitic carbon that is susceptible to being oxidized by reactive oxygen species generated in ORR and have limited oxygen capacities. Here, we report a silica-based electrocatalyst embedding cobalt (Co) sites into silicate-1 (S-1) zeolite, which exhibited exceptional 2e− ORR performance. The aligned microporosity of S-1 zeolite highly isolated Co sites to derive a specific tri-coordination unit of Co(OSi)3 moiety. The nanochannels of S-1 zeolite further provide robust and substantial reaction spaces, where the Co(OSi)3 moiety facilitates O2 chemisorption and holds moderate bonding with *OOH intermediates to reduce the barrier of H2O2 formation. It achieved high Faradaic efficiency over 96% and a steady yield of 11.56 mol gcat−1 h−1 in a gas diffusion electrode setup. This work provides a novel insight into 2e− ORR on Co sites and highlights the potential of zeolite-based materials for electrochemical applications.
Article information: //doi.org/10.1002/anie.202506390